About Iron Deficiency Anemia
What is iron deficiency anemia (IDA)?
Iron deficiency anemia occurs when the level of healthy red blood cells in your body decreases. Red blood cells carry oxygen to the body's tissues. Without enough iron, your body can't produce enough hemoglobin. Hemoglobin is the substance in red blood cells that enables them to carry oxygen.
IDA is the most common type of anemia.
- There are other kinds of anemia, but IDA is the most common, affecting approximately 5 million people in the US
- We get iron from the foods we eat, and it is absorbed into our blood in our small intestine. But if you aren’t getting enough iron, or you’re losing too much iron, or your body isn’t able to absorb it, you may become iron deficient
Common causes of IDA
What are some of the symptoms of IDA?
Symptoms of IDA can be similar to those of other conditions you may have. That’s why it may be helpful to understand what the symptoms of IDA are so you can talk to your healthcare professional about any you may be experiencing.*
Some of the most common symptoms are:
Some people with IDA experience a range of symptoms, while other people experience no symptoms at all. Not all patients feel the same way with IDA. It’s possible to confuse the signs of IDA with symptoms of other conditions you may have.
How is IDA diagnosed?
Only your healthcare professional can diagnose IDA and they will use specific blood tests to diagnose it. IDA is determined by testing three different parts of your blood: hemoglobin (Hb), ferritin, and transferrin saturation (TSAT). It may also be important to test for total iron-binding capacity (TIBC) to diagnose
If you do have IDA, your healthcare professional will continue to use blood tests to keep an eye on your iron levels during treatment.
Learn more about common tests for diagnosing IDA:
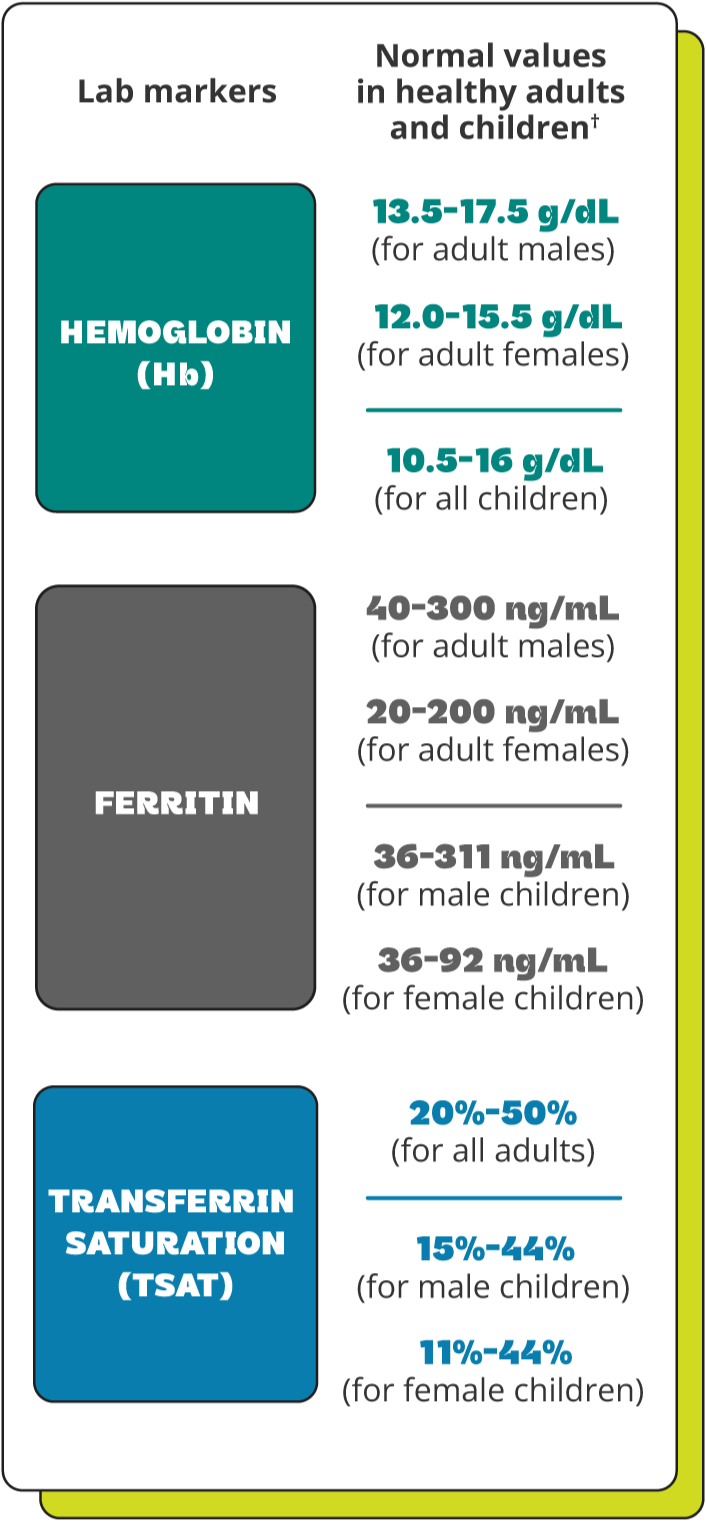
Understanding your lab values
After your healthcare professional orders a blood test, you will receive the lab results and you’ll likely want to know more about what they mean. This chart can help you understand. The chart shows “normal” values, but keep in mind that these can vary for many reasons, depending on the person, medical condition(s), and where the test was taken.
If your lab values are below normal range, talk to your healthcare professional about the best option for restoring your iron levels.

Treatment options for IDA
If your healthcare professional has diagnosed you with iron deficiency anemia (IDA), they may recommend treatment to increase your iron levels. Below are some treatment options. When treating IDA, both oral iron and IV iron are common treatment options.
-

About Oral iron
Learn more about oral iron

Your healthcare professional will most likely start treating your IDA by prescribing an oral iron treatment.
However, there are several reasons why oral iron may not be the right option for some people.
Poor absorption
The digestive tract is only able to absorb a small portion of the iron in an oral iron supplement, so your body may not get the full dose of iron needed.

Even in healthy patients, less than
10% of oral iron is absorbed*Side Effects
Oral iron supplements can cause hard-to-tolerate side effects.
If you have IDA and oral iron supplements aren’t working well enough, ask your healthcare professional about IV iron.
*Oral iron is typically taken in 300 mg or 320 mg tablets 3-4 times a day. The body is unable to absorb that much iron in the digestive tract at one time, so iron repletion may be possible in smaller oral iron doses over time.
-

About IV iron
Learn more about IV iron

With IV treatments, iron is delivered directly into the bloodstream through a vein. Since it doesn’t have to go through your digestive system, 100% of IV iron is delivered into the bloodstream.
If oral iron is inadequate or it causes hard-to-tolerate side effects, IV iron, which is given through an infusion, may be an option. Together, you and your healthcare professional will decide which treatment is best for you.

With IV iron, 100% of iron
is delivered into the bloodstream
How do I know if I’m getting enough iron with my current treatment?
I’m already taking an iron treatment. Why is my Hb level not at goal?
- If you’re taking oral iron supplements: even in healthy people, less than 10% of the iron in oral iron supplements is absorbed.‡ With IV iron, an infusion delivers 100% of iron directly into the bloodstream through a vein
- If you’re on an IV iron treatment: even if you’ve had IV iron infusions, your iron levels may not be where they need to be. Talk to your healthcare professional about how to replenish your iron levels and which IV iron treatment may help
Get help talking about IV Iron with your HEALTHCARE PROFESSIONAL.
Use this tool to create a personalized discussion guide for your next appointment with a healthcare professional.
*Injectafer is not indicated to treat symptoms of IDA.
†Normal lab values may vary based on patient characteristics, comorbidities, and by laboratory.
‡Oral iron is typically taken in 300 mg or 320 mg tablets 3-4 times a day. The body is unable to absorb that much iron in the digestive tract at one time, so iron repletion may be possible in smaller oral iron doses over time.








